Lewis Diagram For Hcn
Jul 26, The Lewis structure for hydrogen cyanide is The steric number for the carbon atom in HCN is 2 and the bonded atom lone pair arrangement. Lewis structure of HCN.

Step method to draw lewis structure of Hydrogen cyanide. Step 1: Find valence e- for all atoms.

Add them together. H C N Lewis structure example. Draw the Lewis Structure of HCN.

1. Draw the skeletal structure showing how the atoms are connected using single bonds. Usually try.

Jul 4, Learn to draw the Lewis structure of HCN & understand molecular geometry, shape, & polarity about the same by reading this article. Refer this.

Answer to Draw the Lewis structure for HCN. Include lone pairs.
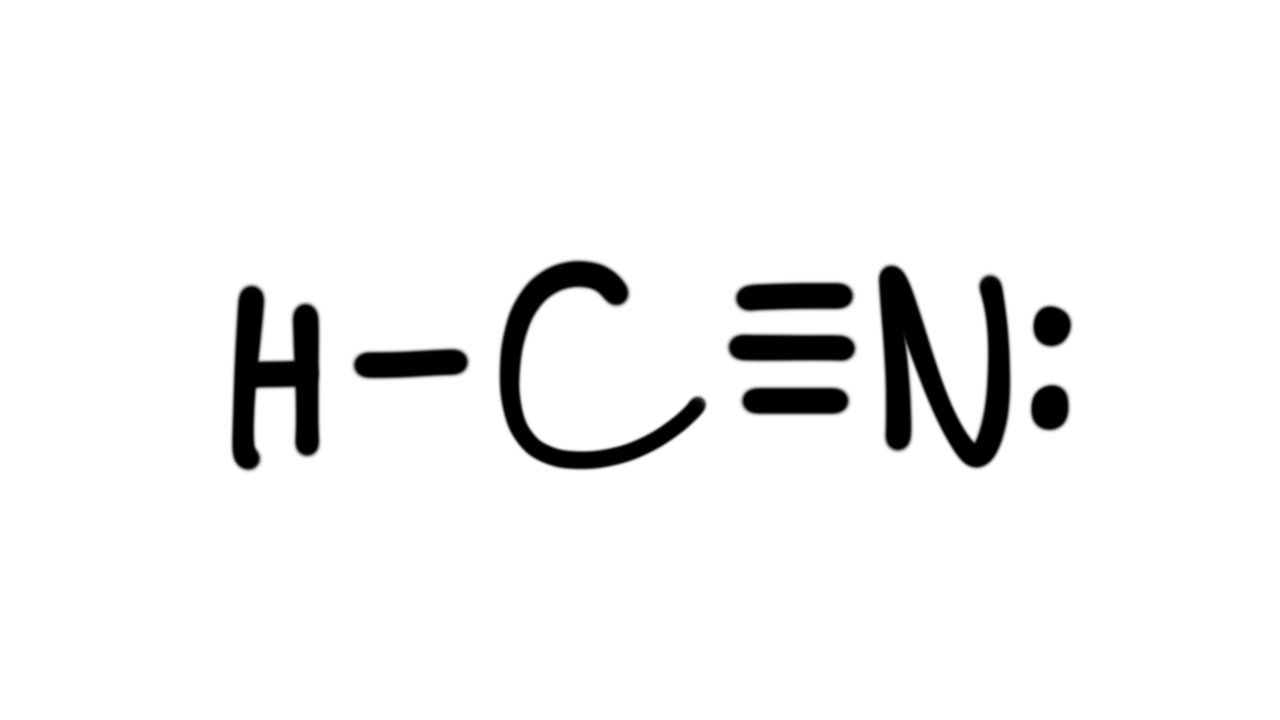
Thanks.A Lewis structure is a graphic representation of the electron distribution around atoms. The reason for learning to draw Lewis structures is to predict the number . views around the world You can reuse this answer Creative Commons License.

Sep 10, · HCN should be written H-CN, with a triple bond between C and N. Observe that the ion CN(-) is isoelectronic with the molecule of CO, carbon monoxide. If you count the valence electrons of CO, you will find 4 for C, and 6 for O, which makes 10 electrons wiringall.com: Resolved.

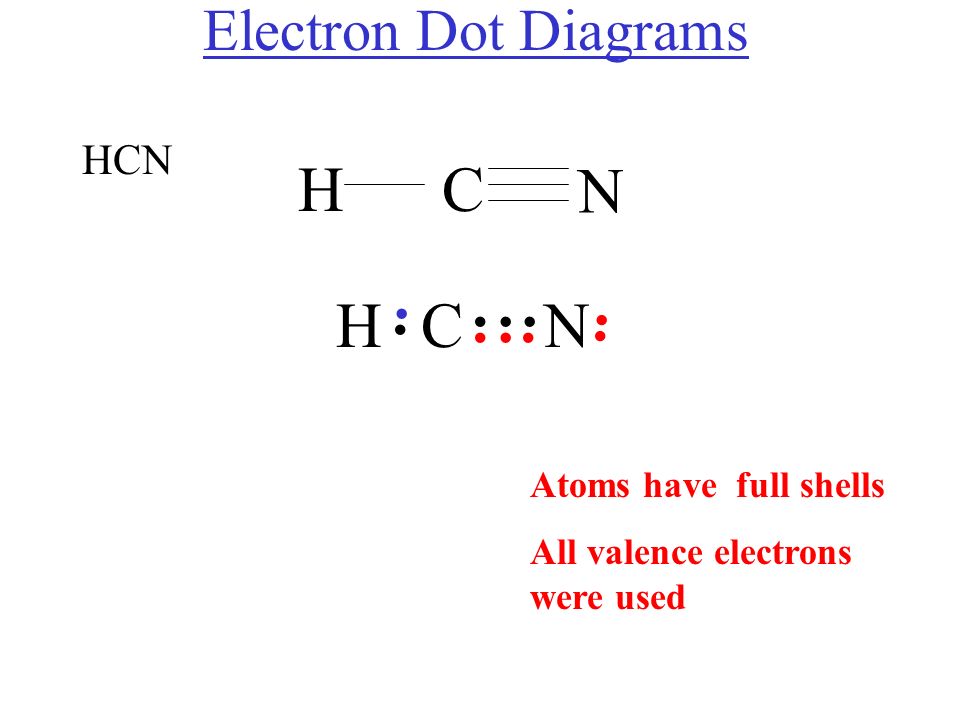
Drawing the Lewis Structure for HCN. Viewing Notes: Make sure you put the correct atom at the center of the HCN molecule.

With the Lewis Structure for HCN you’ll need to share more than one pair of electrons between the Carbon and the Nitrogen atoms. The Lewis structure for HCN, otherwise known as hydrogen cyanide, is fairly simple.

Place the carbon atom in the center and triple bond it to a nitrogen atom. Then bond the carbon atom to a single hydrogen atom. The nitrogen atom will have a lone pair placed on it.Linear Molecular Geometry – Chemistry LibreTextsHCN Lewis Structure [w/ free video guide]
